
Some copy here
more copy
Resources
Personalized Medicine 101
The Story
Personalized medicine uses new methods of molecular analysis to better manage a patient’s disease or predisposition toward a disease. It aims to achieve optimal medical outcomes by helping physicians and patients choose the disease management approaches likely to work best in the context of a patient’s genetic and environmental profile. Such approaches may include genetic screening programs that more precisely diagnose diseases and their sub-types, or help physicians select the type and dose of medication best suited to a certain group of patients.
People vary from one another in many ways — what they eat, the types and amount of stress they experience, exposure to environmental factors, and their DNA. Many of these variations play a role in health and disease. For example, the natural variations found in our genes could influence our risk of developing a certain disease, as well as how our bodies respond to that disease. The combination of these variations across several genes can affect each individual’s risk of developing a disease or reacting to something in the environment, and can be one of the reasons why a drug works for one patient and not another.
Personalized medicine hopes to use these variations — both in the patient and in the molecular underpinnings of the disease itself — to develop new treatments and to identify the sub-groups of patients for whom they will work best. It can also help determine which groups of patients are more prone to developing some diseases and, ideally, help with the selection of lifestyle changes and/or treatments that can delay onset of a disease or reduce its impact.
Personalized medicine is poised to transform healthcare over the next several decades. New diagnostic and prognostic tools will increase our ability to predict the likely outcomes of drug therapy, while the expanded use of biomarkers — biological molecules that indicate a particular disease state — could result in more focused and targeted drug development. Personalized medicine also offers the possibility of improved health outcomes and has the potential to make healthcare more cost-effective.
Though sometimes described as a phenomenon of the future, personalized medicine is already having an impact on how patients are treated. Molecular testing is being used to identify those breast cancer and colon cancer patients likely to benefit from new treatments, and newly diagnosed patients with early stage invasive breast cancer can now be tested for the likelihood of recurrence. In another example, a genetic test for patients with an inherited cardiac condition can help their physicians determine which course of hypertension treatment to prescribe in order to avoid serious side effects.
Personalized medicine promises many medical innovations, and has the potential to change the way treatments are discovered and used. But the pathway to the development of personalized medicine is marked by the need to identify and address a range of public policy issues. The implications for current systems, such as payer and physician incentives, medical records privacy and clinical trial ethics, must be explored by all stakeholders, who will need to reach agreement on what modifications should be made. The way such issues are managed will affect the evolution of personalized medicine and shape its ability to prevent, diagnose and manage disease.
The paradigm of personalized medicine can be illustrated as follows:
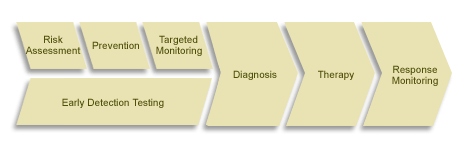
This arrow reflects the current and anticipated flow of health care services, and changing points of intervention, as medicine becomes more personalized. Early detection testing will continue based on large population risk (e.g., mammograms), while new forms of risk assessment will be incorporated (e.g., determining which women carry the genetic variation that increases their risk for developing cancer). Though true prevention must occur before disease symptoms are present, better risk assessment enables more targeted monitoring (e.g., women with the genetic variation should have more frequent mammograms); followed by symptom-driven diagnosis, in which molecular monitoring could possibly identify disease subtypes that cannot be clinically determined. Such diagnosis may or may not lead to targeted therapy, but in either event we may also benefit from improvements in monitoring a patient’s response to a particular therapy.
Read about the science behind personalized medicine. >>

